Automating Cell Therapy Manufacturing
Chronicle is a GMP-compliant, fit-for-purpose digital solution developed by Cytiva to optimize complex cell therapy process development and manufacturing.
It connects Cytiva and third-party cell therapy instruments through a unified platform, supporting the full journey from early process development through commercial GMP manufacturing.
The platform provides electronic batch records, instrument scheduling, deviation management, and supply chain traceability - all independently audited against GAMP5 standards and compliant with 21 CFR Part 11 (FDA) and EU Annex 11 (EMA).
My role
I contributed to the front-end development of Chronicle, building key interfaces for instrument management, facility monitoring, and electronic standard operating procedures (eSOPs).
Unified Dashboard
One central view for your entire manufacturing operation
Chronicle’s home dashboard aggregates real-time data across all connected instruments, customer trials, pre-GMP and GMP organisations, and remaining credit usage - giving operations teams instant situational awareness.
Instruments Overview
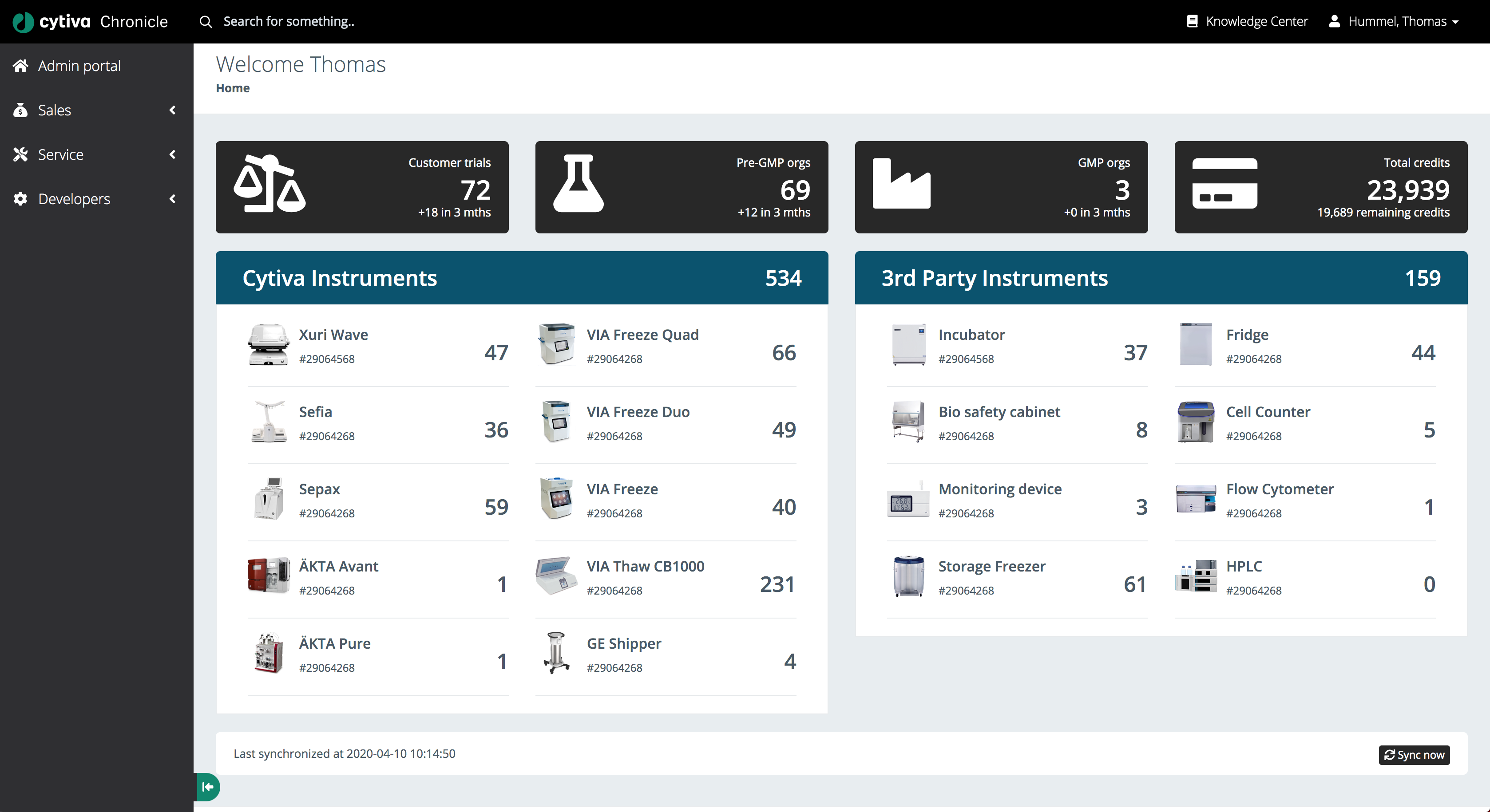
The main dashboard displays all connected instruments across two categories:
- Cytiva Instruments: Xuri Wave, Sofia, Sepax, AKTA Avant, AKTA Pure, VIA Freeze Quad, VIA Freeze Duo, VIA Freeze, VIA Thaw CB1000, GE Shipper
- 3rd Party Instruments: Incubator, Fridge, Bio safety cabinet, Cell Counter, Monitoring device, Flow Cytometer, Storage Freezer, HPLC
Each instrument card shows its current unit count in real time. The header bar provides a summary of customer trials, pre-GMP organisations, GMP organisations, and total credits remaining - giving managers an instant overview of capacity.
Equipment List
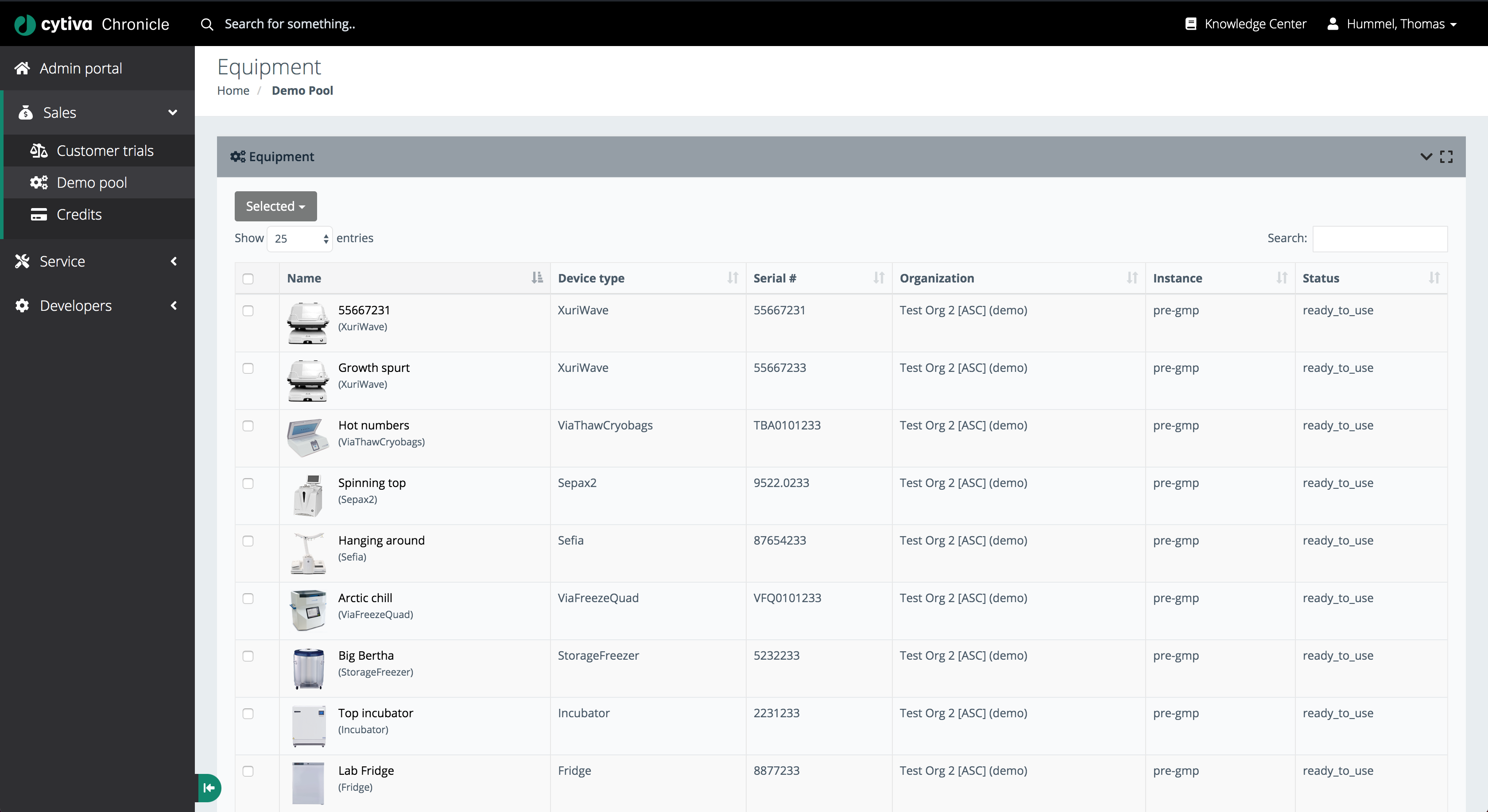
The equipment list provides a sortable, searchable table of every registered instrument across all demo pools and organisations. Each row surfaces:
- Instrument name and nickname
- Device type
- Serial number
- Organisation and instance
- Current status (e.g.
ready_to_use)
This centralised registry eliminates spreadsheet-based asset tracking and provides a single source of truth for all lab instruments.
Equipment Details and Connectivity
Full instrument traceability with remote access capabilities
Each instrument record captures cumulative runtime, data points, service history, and live alarm counts - along with direct links to ServiceMax and remote login access.
Instrument Detail View
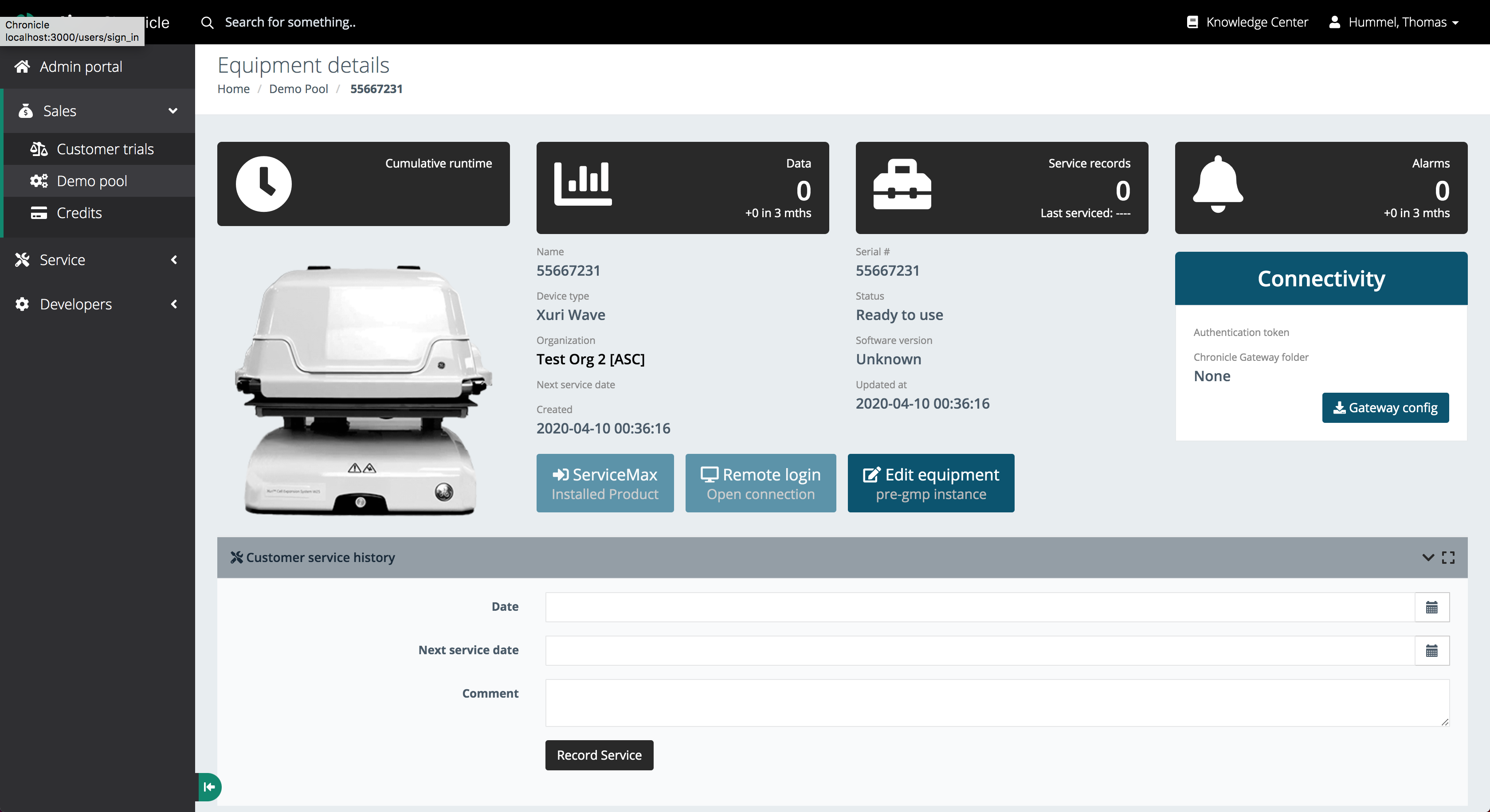
The equipment detail page aggregates all data related to a single instrument:
- Cumulative runtime counter
- Data points collected
- Service records with last serviced date
- Alarms with delta over the last 3 months
- Serial number, device type, organisation, software version, and creation date
Operators can trigger a ServiceMax ticket, open a Remote Login session to the instrument directly, or switch the instance to GMP mode - all from the same screen.
A Connectivity panel shows authentication tokens and the Chronicle Gateway folder path, making it easy to diagnose integration issues without leaving the UI.
Work Queue and Activity Stream
Coordinating multi-step workflows across teams and instruments
The work queue surfaces all in-progress and pending eSOPs alongside a live activity stream, so nothing falls through the cracks between shifts.
Real-time Operations View
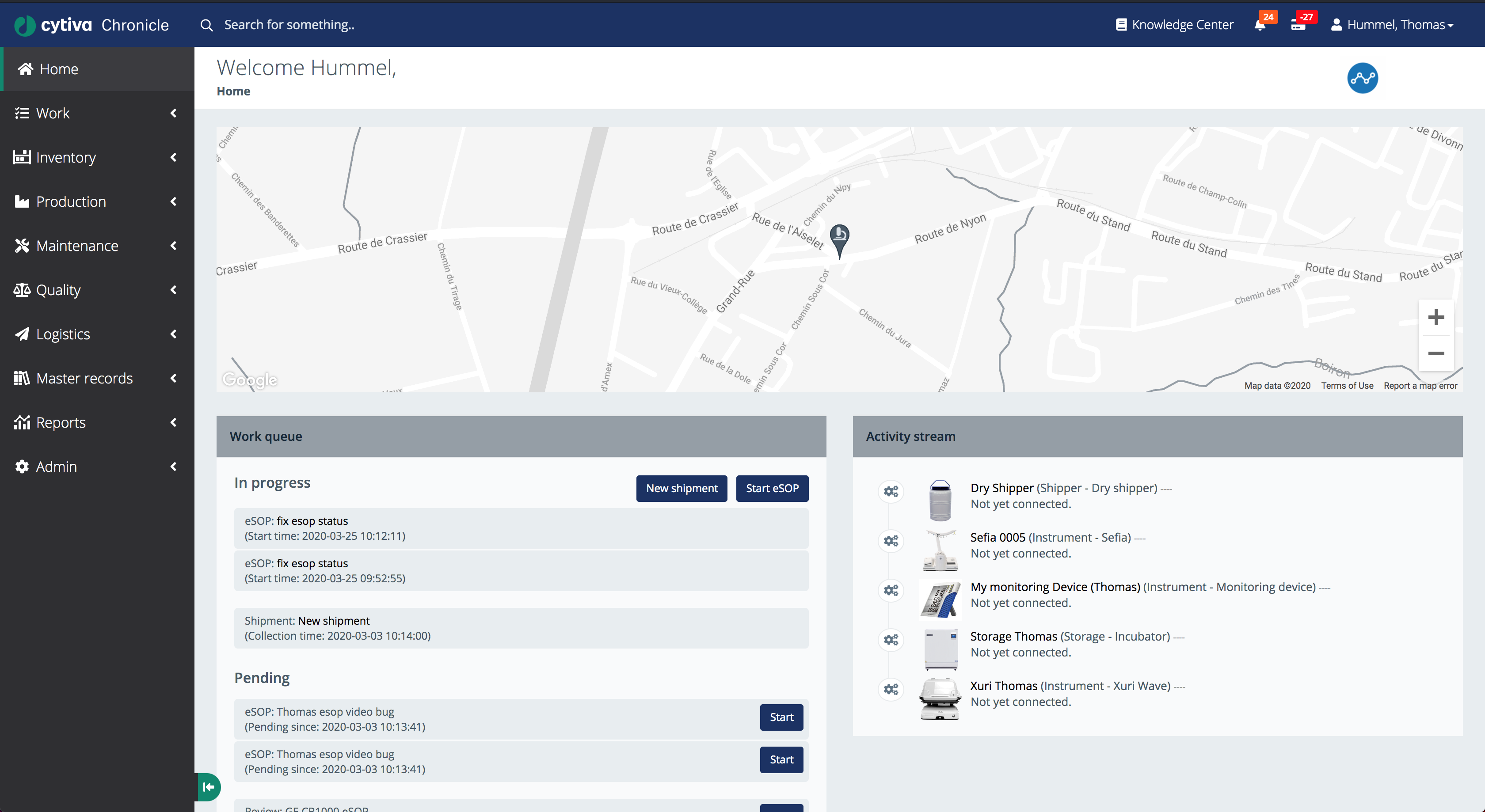
The home screen for GMP operators combines three panels:
- Map view: displays the physical location of the manufacturing facility, providing geographical context for multi-site deployments
- Work queue: lists all active and pending eSOPs with start dates, allowing operators to pick up tasks and track progress
- Activity stream: a live feed of instrument connection events and status changes across all registered devices
New shipments can be initiated directly from this screen, and eSOPs can be started with a single click - reducing the friction between planning and execution.
Facility Dashboard
A visual floor plan of your manufacturing environment
The facility dashboard overlays real-time instrument data onto a floor plan, making it immediately clear which instruments are in alert state and where they are physically located.
Floor Plan Monitoring
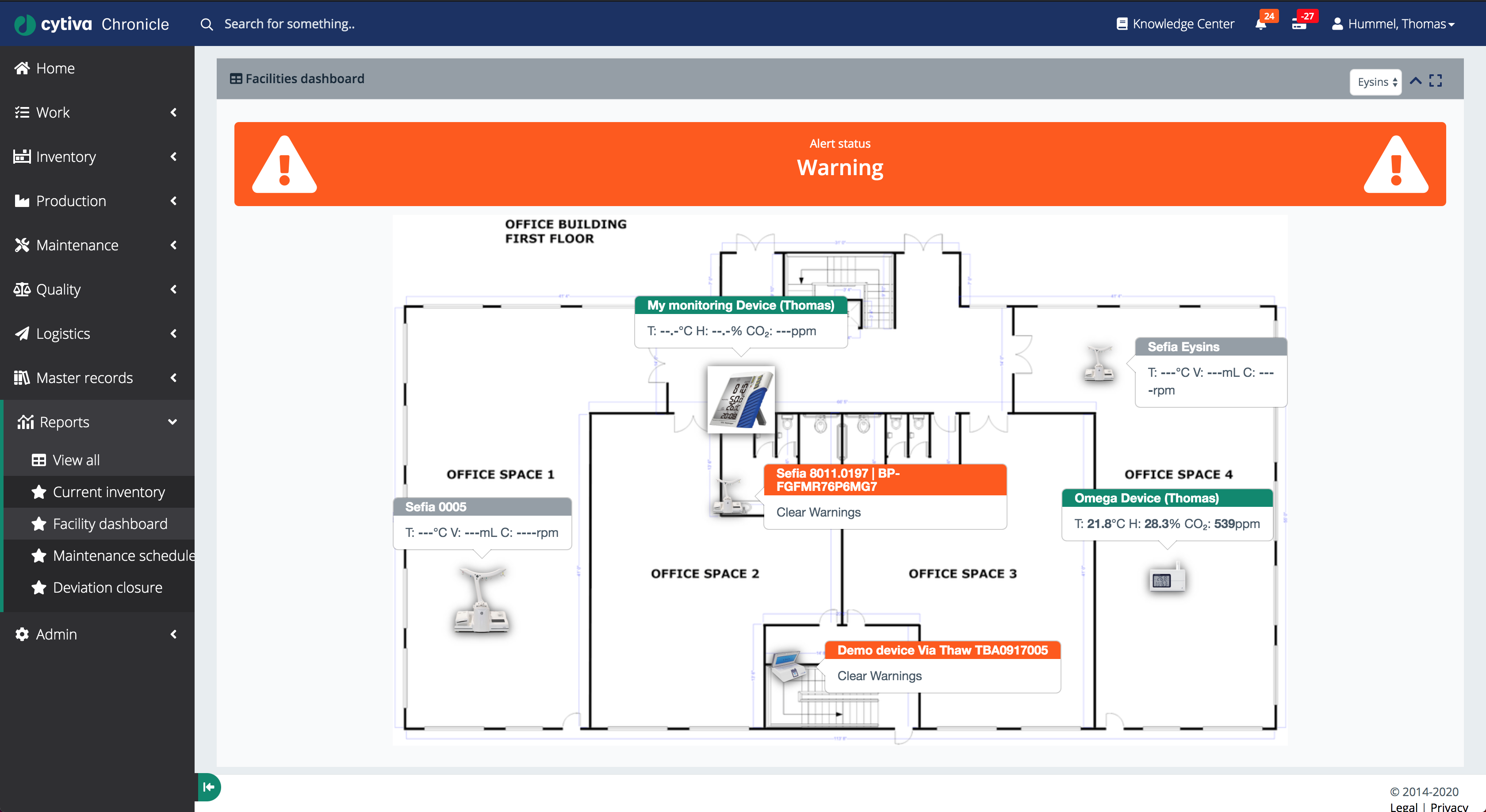
The facility dashboard renders a floor plan of the manufacturing space with each connected instrument pinned to its physical location. Instruments in a warning state are highlighted in orange with a warning banner at the top of the screen.
Each pin displays live sensor readings (temperature, CO₂ levels) and current status, allowing facility managers to spot environmental deviations at a glance without walking the floor.
This view integrates with the alarm and deviation management system, so clicking a warning pin opens the relevant deviation workflow directly.
Electronic Standard Operating Procedures (eSOPs)
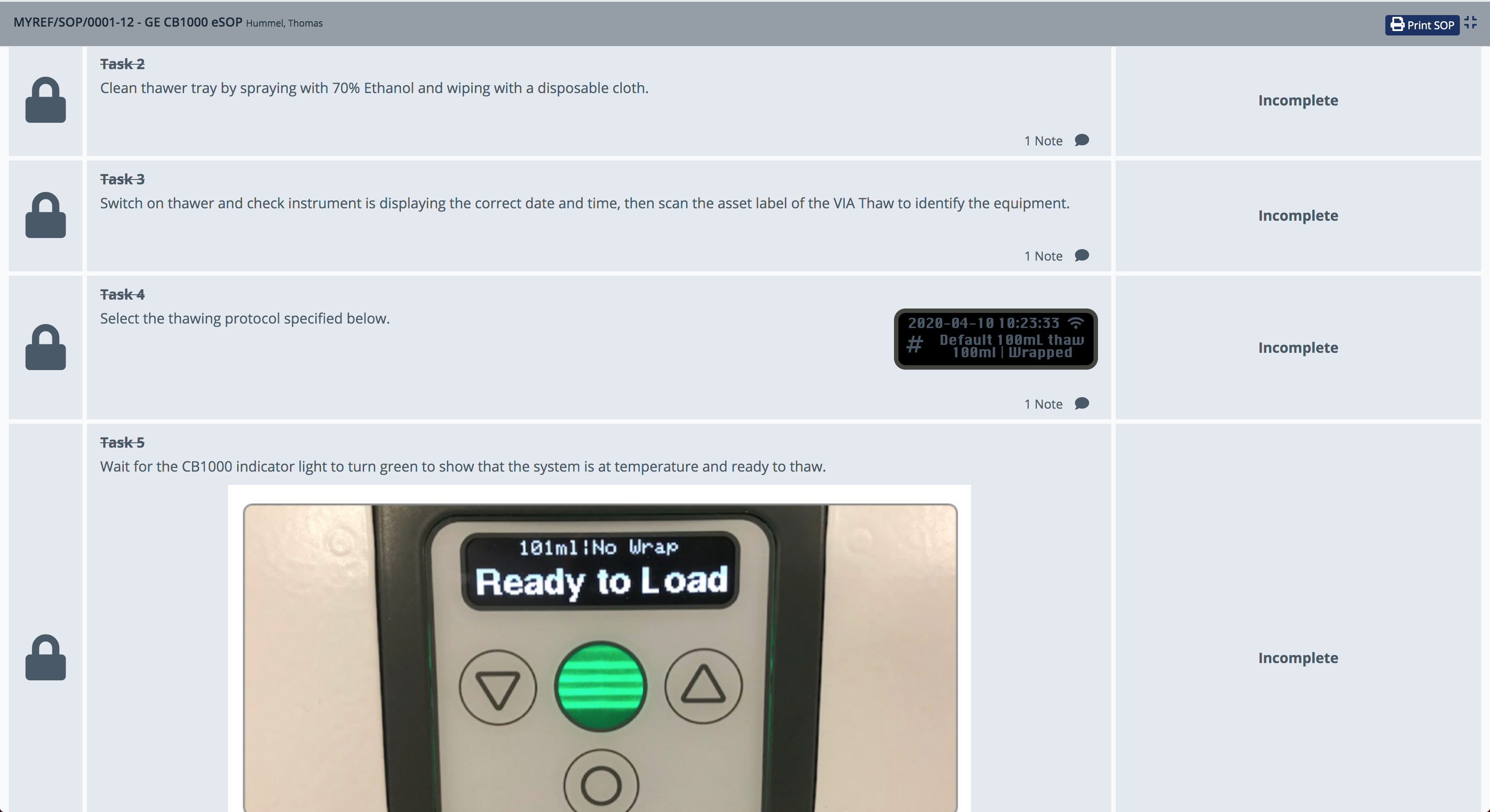
Chronicle replaces paper SOPs with guided, digital step-by-step procedures. Each eSOPs task:
- Locks steps sequentially to enforce correct execution order
- Embeds photographic references of instrument displays to guide operators
- Records completion timestamps and operator identity for audit trail
- Supports deviation notes per task
This approach ensures every operator follows the exact same procedure every time, regardless of experience level, and produces a complete electronic record compliant with GMP requirements.
Chronicle, it is
- A GMP-compliant Manufacturing Execution System (MES) for cell therapy
- Electronic batch records audited against GAMP5
- Compliance with 21 CFR Part 11 and EU Annex 11
- Integration with Cytiva and third-party instruments
- Instrument scheduling, deviation management, and supply chain traceability
- Real-time facility monitoring via interactive floor plans
- Guided eSOPs with full audit trail
Technology: Ruby on Rails / React / PostgreSQL / Docker / REST APIs